Safety
Adverse Reactions
Many of the adverse reactions in the LUTATHERA arm were grade 1 or 2.1
Adverse Reactions Occurring at a Higher Incidence in the LUTATHERA Arm (Between-Arm Difference of ≥5% [All Grades] or ≥2% [Grade 3/4])1 | ||||
| LUTATHERA With Long-Acting | Long-Acting | ||
Adverse Reactiona | All | Grade | All | Grade |
Gastrointestinal disorders | ||||
Nausea | 65 | 5 | 12 | 2 |
Vomiting | 53 | 7 | 10 | 0 |
Abdominal pain | 26 | 3 | 19 | 3 |
Diarrhea | 26 | 3 | 18 | 1 |
Constipation | 10 | 0 | 5 | 0 |
General disorders | ||||
Fatigue | 38 | 1 | 26 | 2 |
Peripheral edema | 16 | 0 | 9 | 1 |
Pyrexia | 8 | 0 | 3 | 0 |
Metabolism and nutrition disorders | ||||
Decreased appetite | 21 | 0 | 11 | 3 |
Nervous system disorders | ||||
Headache | 17 | 0 | 5 | 0 |
Dizziness | 17 | 0 | 8 | 0 |
Dysgeusia | 8 | 0 | 2 | 0 |
Vascular disorders | ||||
Flushing | 14 | 1 | 9 | 0 |
Hypertension | 12 | 2 | 7 | 2 |
Musculoskeletal and connective tissue disorders | ||||
Back pain | 13 | 2 | 10 | 0 |
Pain in extremity | 11 | 0 | 5 | 0 |
Myalgia | 5 | 0 | 0 | 0 |
Neck pain | 5 | 0 | 0 | 0 |
Renal and urinary disorders | ||||
Renal failureb | 13 | 3 | 4 | 1 |
Radiation-related urinary tract adverse reactionsc | 8 | 0 | 3 | 0 |
Psychiatric disorders | ||||
Anxiety | 12 | 1 | 5 | 0 |
Skin and subcutaneous tissue disorders | ||||
Alopecia | 12 | 0 | 2 | 0 |
Respiratory, thoracic, and mediastinal disorders | ||||
Cough | 11 | 1 | 6 | 0 |
Cardiac disorders | ||||
Atrial fibrillation | 5 | 1 | 0 | 0 |
aNational Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE) Version 4.03 only displays adverse reactions occurring at a higher incidence in patients treated with LUTATHERA (between-arm difference of ≥5% [all grades] or ≥2% [grade 3/4]).
bIncludes the terms glomerular filtration rate decreased, acute kidney injury, acute prerenal failure, azotemia, renal disorder, renal failure, and renal impairment.
cIncludes the terms dysuria, micturition urgency, nocturia, pollakiuria, renal colic, renal pain, urinary tract pain, and urinary incontinence.
Adverse events that were considered by the investigator to be related to trial treatment occurred in 129 patients.2
95 patients (86%) in the LUTATHERA arm and 34 patients (31%) in the control arm2
Premature withdrawal due to adverse reactions was more common in patients treated with octreotide alone vs LUTATHERA with octreotide (10 patients or 9% vs 7 patients or 6%, respectively)
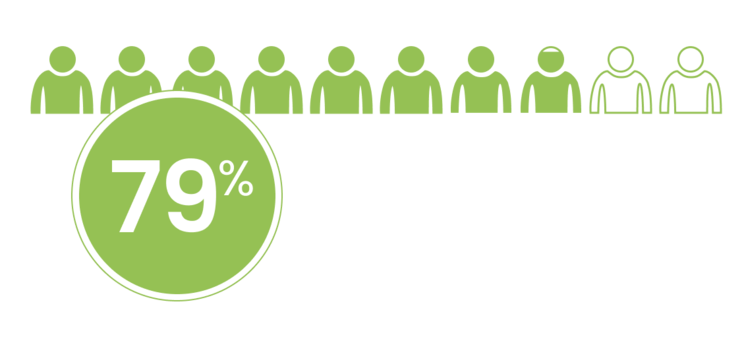
of LUTATHERA patients received a cumulative dose >22.2 GBq (>600 mCi) in NETTER-1.1
Laboratory Abnormalities
Laboratory Abnormalities Occurring at a Higher Incidence in the LUTATHERA Arm (Between-Arm Difference of ≥5% [All Grades] or ≥2% [Grade 3/4])1,d,e | ||||
| LUTATHERA With Long-Acting | Long-Acting | ||
Laboratory Abnormalityd,e | All | Grade | All | Grade |
Hematology | ||||
Lymphopenia | 90 | 44 | 39 | 5 |
Anemia | 81 | 0 | 55 | 1 |
Leukopenia | 55 | 2 | 20 | 0 |
Thrombocytopenia | 53 | 1 | 17 | 0 |
Neutropenia | 26 | 3 | 11 | 0 |
Renal/metabolic | ||||
Creatinine increased | 85 | 1 | 73 | 0 |
Hyperglycemia | 82 | 4 | 67 | 2 |
Hyperuricemia | 34 | 6 | 30 | 6 |
Hypocalcemia | 32 | 0 | 14 | 0 |
Hypokalemia | 26 | 4 | 21 | 2 |
Hyperkalemia | 19 | 0 | 11 | 0 |
Hypernatremia | 17 | 0 | 7 | 0 |
Hypoglycemia | 15 | 0 | 8 | 0 |
Hepatic | ||||
GGT increased | 66 | 20 | 67 | 16 |
Alkaline phosphatase increased | 65 | 5 | 55 | 9 |
AST increased | 50 | 5 | 35 | 0 |
ALT increased | 43 | 4 | 34 | 0 |
Blood bilirubin increased | 30 | 2 | 28 | 0 |
dValues are worst grade observed after randomization.
eNational Cancer Institute Common Technology Criteria for Adverse Events (CTCAE) Version 4.03 only displays laboratory abnormalities occurring at a higher incidence in patients treated with LUTATHERA (between-arm difference of ≥5% [all grades] or ≥2% [grade 3/4]).
Dose reduction and discontinuation
6% (7 of 111) of patients required a dose reduction, and 13% (14 of 111) of patients discontinued LUTATHERA1
– 5 patients discontinued due to renal-related events– 4 patients discontinued due to hematologic toxicities
The most common grade 3/4 adverse reactions with a higher incidence in the LUTATHERA arm were lymphopenia (44%), increased GGT (20%), vomiting (7%), nausea (5%), increased AST (5%), increased ALT (4%), hyperglycemia (4%), and hypokalemia (4%).1
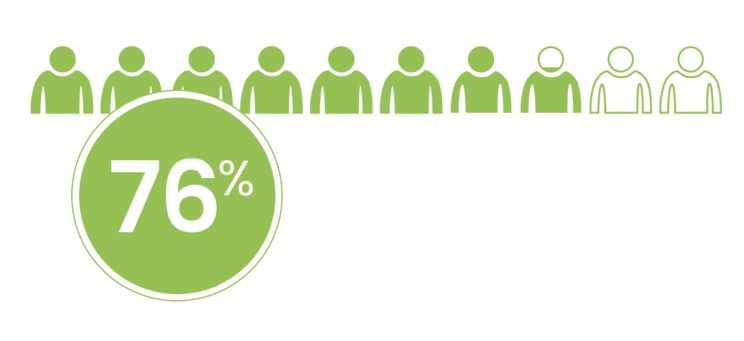
of patients treated with LUTATHERA and long-acting octreotide received all 4 planned doses in NETTER-1.1
NETTER-1 Final Analysis
No new safety signals were reported in the 5-year, long-term follow-up3,f
Adverse Events | During the long-term follow-up, only serious adverse events (SAEs) deemed related to treatment with LUTATHERA and AEs of special interest (hematotoxicity, cardiovascular events, and nephrotoxicity, regardless of causality) in the LUTATHERA arm were reported3 |
Grade ≥3 Treatment-Related SAEs During the Entire Study | 7 (6%) of 111 patients treated in the LUTATHERA arm3 |
Incidence of Treatment-Related SAEs During the Long-Term Follow-Up Period | 3 (3%) of 111 patients treated with LUTATHERA3
|
MDS or Acute Leukemia | No new cases were reported during long-term follow-up3
|
Diffuse Large B-Cell Lymphoma | One patient developed diffuse large B-cell lymphoma during long-term follow-up that was deemed unrelated to treatment with LUTATHERA3 |
Nephrotoxicity of Grade ≥3, Regardless of Causality | Reported in 6 (5%) of 111 patients in the LUTATHERA arm and 4 (4%) of 112 patients in the control arm during the study3 |
fCutoff date for final analysis was January 18, 2021.3
ERASMUS: Long-Term Safety
ERASMUS, A RETROSPECTIVE, LONG-TERM (MEDIAN FOLLOW-UP, >4 YEARS), OPEN-LABEL TRIAL | |
STUDY DESIGN | Retrospective safety data are available from 1214 patients in ERASMUS, an international, single-institution, single-arm, open-label trial of patients with SSTR-positive tumors (neuroendocrine and other primaries).1 |
ADMINISTRATION | LUTATHERA 7.4 GBq (200 mCi) was administered every 6 to 13 weeks for up to 4 doses with or without octreotide. Retrospective medical record review was conducted on a subset of 811 patients to document serious adverse reactions.1
|
Serious adverse reactions1
patients developed | patients developed | ||
|
| ||
- Hepatotoxicity was also observed in ERASMUS, with 2 patients (<1%) reported to have hepatic tumor hemorrhage, edema, or necrosis, and 1 patient experiencing intrahepatic congestion and cholestasis. Patients with hepatic metastasis may be at increased risk of hepatotoxicity due to radiation exposure1
- Monitor transaminases, bilirubin, serum albumin, and the international normalized ratio (INR) during treatment. Withhold dose, reduce dose, or permanently discontinue LUTATHERA based on the severity of hepatotoxicity
Please see warnings and precautions for myelosuppression, MDS, leukemia, renal toxicity, hypersensitivity reactions, and neuroendocrine hormonal crisis. Monitor blood cell counts. Withhold dose, reduce dose, or permanently discontinue based on severity of adverse reactions.1
Warnings and Precautions
Events Reported in the NETTER-1 and ERASMUS Clinical Trials
RISK FROM RADIATION EXPOSURE |
|
MYELOSUPPRESSION |
|
SECONDARY MDS AND LEUKEMIA |
|
RENAL TOXICITY |
|
HEPATOTOXICITY |
|
HYPERSENSITIVITY REACTIONS |
|
NEUROENDOCRINE HORMONAL CRISIS |
|
EMBRYO-FETAL TOXICITY |
|
RISK OF INFERTILITY |
|
Radiation Safety
Important Safety Instructions1
LUTATHERA is a radiopharmaceutical; handle with appropriate safety measures to minimize radiation exposure. Use waterproof gloves and effective radiation shielding when handling LUTATHERA. Radiopharmaceuticals, including LUTATHERA, should be used by or under the control of health care professionals who are qualified by specific training and experience in the safe use and handling of radiopharmaceuticals, and whose experience and training have been approved by the appropriate governmental agency authorized to license the use of radiopharmaceuticals. Verify pregnancy status of females of reproductive potential prior to initiating LUTATHERA.
Monitor patients closely for signs and symptoms of hypersensitivity reactions during and following the LUTATHERA administration for a minimum of 2 hours in a setting in which cardiopulmonary resuscitation medication and equipment are available.
ALARA Principle: Keeping Radiation Exposure as Low as Reasonably Achievable4
ALARA is the guiding principle of radiation safety. It means that even a small radiation dose should be avoided if there is no benefit to receiving it. It includes 3 basic protective measures:
| Minimize time near a radiation source. Spend only the time needed to complete your job near the radiation source and then leave the area. |
| Maximize distance from a radiation source. Stay as far away as you can from the radiation source. |
| Use appropriate shielding between yourself and a radiation source. Put appropriate materials between you and the radiation source. The appropriate materials will depend on what type of radiation the source emits. |
Safety Procedures
You should follow these procedures in addition to your institution’s radiation safety guidelines whenever handling or administering LUTATHERA:
General handling and administration recommendations
Use waterproof gloves1,5
Use aseptic technique and radiation shielding1
Dispose of any unused medicinal product or waste material in accordance with local and federal laws1
Additional handling and administration recommendations
Wear gowns and shoe covers1,5
Use tongs when handling vial to minimize radiation exposure1
Radiation Exposure
Radiation Associated With LUTATHERA
TYPES OF RADIATION EMITTED | LUTATHERA decays to stable hafnium (Hf-177), with a half-life of 6.647 days, by emitting beta minus (β-) radiation with a maximum energy of 0.498 MeV (79%) and photonic radiation (γ) of 0.208 MeV (11%) and 0.113 MeV (6.2%)1 |
PENETRATING RADIATION | The maximum radiation penetration in tissue is 2.2 mm, and the mean penetration is 0.67 mm1 |
PATIENT EXPOSURE | Patients are discharged from the treatment center only when radiation exposure to third parties does not exceed regulatory thresholds5 |
Radiation Exposure to HCPs and Caregivers Following Outpatient Treatment With Lutetium-177
Methods
Seventy-six patients with progressive, metastatic NETs received 4 cycles of 7.8 GBq of LUTATHERA at 8-week intervals in an outpatient setting at 1 treatment center. Four patients were treated sequentially on each therapy day in a 4-bed room in the hospital’s day procedure unit, with each patient remaining until radiation exposure was below the release limit. Radiation exposure to HCPs and caregivers was monitored by personal dosimeter.6
Radiation study results
HEALTH CARE PROFESSIONALS | Mean whole-body exposures per therapy treatment day with 4 patients when administering LUTATHERA ranged from 6.8 μSv (nuclear medicine technologist) to 33.2 μSv (nurse). In the nearby staff office with a 50% staff occupancy, the mean exposure rate measured on 10 different therapy administration days was 1.6 μSv/h (range, 1.3–2.0 μSv/h), whereas that at the nursing station with 100% staff occupancy was 3.5 μSv/h (range, 2.9–4.0 μSv/h).6,g |
CAREGIVERS | Mean total exposure during the day of therapy and at home for a period of up to 5 days was 90 µSv, with a median exposure of 40 μSv and range of 10 μSv to 470 μSv.6,g,h |
Exposures to HCPs and caregivers were within the limits recommended by the International Commission on Radiological Protection.6 | |
gFor reference, mean radiation exposure is 14.5 μSv on a 5.2-hour flight from Los Angeles to Honolulu.7
hTwenty-five caregivers were provided with electronic dosimeters.6
Radiation Spill
If a radiation spill occurs, you should always follow the guidance of your institution’s radiation safety department. The information below is guidance from the National Radiation Commission.8
FOR MINOR SPILLS8 | |
NOTIFY | Notify persons in the area that a spill has occurred. |
PREVENT THE SPREAD | Cover the spill with absorbent paper. |
CLEANUP | Use disposable gloves and absorbent paper. Carefully fold the absorbent paper with the clean side out and place in a labeled plastic bag for transfer to a radioactive waste container. Also put contaminated gloves and any other contaminated disposable material in the bag. |
SURVEY | With a low-range radiation detection survey meter, check the area around the spill. Also check your hands, clothing, and shoes for contamination. |
REPORT | Report the incident to the Radiation Safety Officer (RSO). |
DOCUMENT | Complete any necessary forms for documentation. |
FOR MAJOR SPILLS8 | |
CLEAR THE AREA | Notify all persons not involved in the spill to vacate the room. |
PREVENT THE SPREAD | Cover the spill with absorbent paper, but do not attempt to clean it up. To prevent the spread of contamination, limit the movement of all personnel who may be contaminated. |
SHIELD THE SOURCE | If possible, shield the spill. This should be done only if it can be done without further contamination or a significant increase in radiation exposure. |
CLOSE THE ROOM | Lock or otherwise secure the area to prevent entry. |
CALL FOR HELP | Notify the RSO immediately. |
PERSONNEL DECONTAMINATION | Remove contaminated clothing and flush contaminated skin with lukewarm water and then wash with mild soap. If contamination remains, induce perspiration by covering the area with plastic. Then wash the affected area again to remove any contamination that was released by the perspiration. |
CLEANUP | The RSO will supervise the cleanup of the spill and complete any necessary forms for documentation. |
Next: Dosing and Administration
AEs, adverse events; ALT, alanine aminotransferase; AST, aspartate aminotransferase; CrCL, creatinine clearance; GGT, gamma-glutamyl transferase; HCPs, health care professionals; MDS, myelodysplastic syndrome; NETs, neuroendocrine tumors; SSA, somatostatin analog; SSTR, somatostatin receptor.
